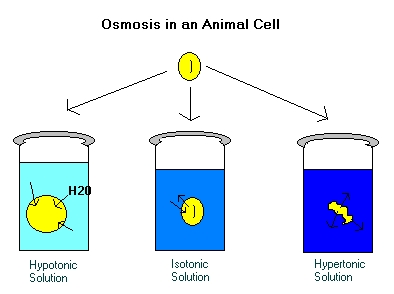
Osmosis is important in animal cells because
it allows them to interact with their surrounding environment. In
the above diagram, the yellow shapes represent animal cells. When
the animal cell is placed in a hypotonic solution, most of the water will
move into the cell, making it grow in size and mass. When the cell
is placed in an isotonic solution, an equal amount of water will move in
and out of the cell, and the size and mass will remain constant..
In a hypertonic solution, most of the water will move out of the cell,
leaving the cell with less mass, and a shriveled form.
Home